Goal 2 - Osmosis
Osmosis
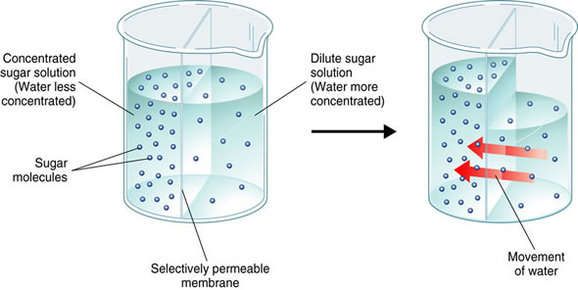
THE DIFFUSION OF WATER ACROSS A SEMIPERMEABLE MEMBRANE IS CALLED OSMOSIS.
THE DIFFUSION OF WATER ACROSS A SEMIPERMEABLE MEMBRANE IS CALLED OSMOSIS.
- THE PROCESS BY WHICH WATER MOLECULES DIFFUSE ACROSS A CELL MEMBRANE FROM AN AREA OF HIGHER CONCENTRATION TO AN AREA OF LOWER CONCENTRATION IS CALLED OSMOSIS.
- Water move across a Cell Membrane from a region of HIGH Concentration of Water to a region of LOW Concentration.
- Like any other Diffusing Molecule, Water moves DOWN its Concentration Gradient.
- SOLUTES ARE DISSOLVED SUBSTANCES IN A SOLUTION. Cytoplasm (jelly-like substance in the cell) is Mostly Water containing many dissolved solutes.
- Because no TWO Molecules can occupy the same space at the same time, the more Solutes there are in a certain volume of Water, The FEWER Water Molecules there can be in the same volume.
- Solutions with many Solutes contain FEWER Water Molecules than do solutions with fewer solutes.
- Water will cross the Membrane toward the Higher Solute Concentration until the Concentration Gradients of BOTH WATER and SOLUTES EVEN OUT.
- The Net Direction of Osmosis DEPENDS on the Relative Concentration of Solutes on the TWO Sides of the Cell Membrane.
What Happens to the Cell?
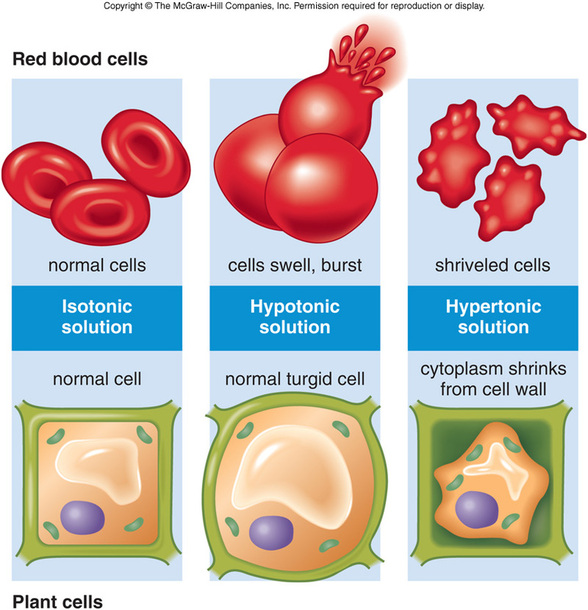
If the cell loses too much water, the cell will shrivel and shrink.
The Flow of water into a cell causes it to swell.
Animal cells placed in distilled water will swell and often burst because of Osmosis.
IN AN SOLUTION, where THE CONCENTRATION OF SOLUTES OUTSIDE AND INSIDE THE CELL ARE EQUAL, water will Diffuse Into and Out of the Cell at EQUAL RATES
The Flow of water into a cell causes it to swell.
Animal cells placed in distilled water will swell and often burst because of Osmosis.
- The Bursting of Cells is called CYTOLYSIS. (sie-TAHL-uh-suhs)
IN AN SOLUTION, where THE CONCENTRATION OF SOLUTES OUTSIDE AND INSIDE THE CELL ARE EQUAL, water will Diffuse Into and Out of the Cell at EQUAL RATES
- So there is NO Net Movement of Water
