Goal 2 - Diffusion Notes
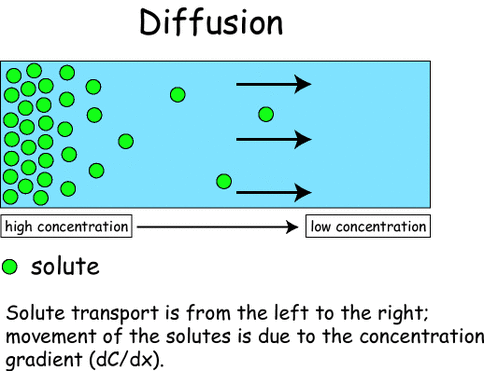
Diffusion
- Diffusion is Driven By the KINETIC ENERGY (energy of motion) the molecules possess.
- Because of their Kinetic Energy, molecules are in Constant Motion.
- Diffusion occurs when molecules move randomly away from each other in a liquid or gas Atoms and molecules exhibit random motion.
- This motion is characteristic of solids, liquids or gases.
- Particles of different substances that are in constant motion have an effect on each other.
- DIFFUSION is the movement of molecules from an area of higher concentration to an area of lower concentration.
- Result of the random movement of particles.
- Does Not require the cell to use Energy, is DIFFUSION.
- Diffusion is the simplest type of PASSIVE TRANSPORT
- Small molecules can pass through the cell membrane by a process called DIFFUSION.
Rates of Dissusion
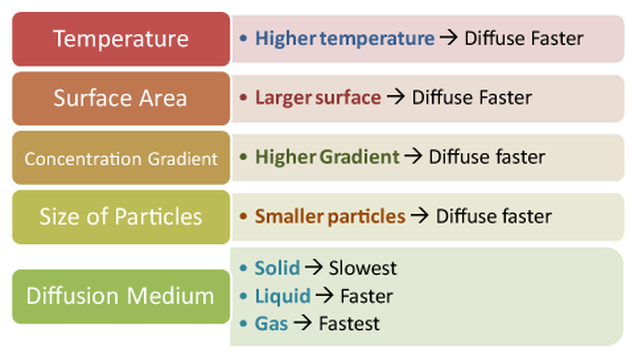
The Rate of Diffusion depends on the:
The more CONCENTRATED the substances, the more rapidly diffusion occurs because there are more collisions between the particles of a substances.
Molecules Diffuse FASTER at HIGHER TEMPERATURES than at lower temperatures.
SMALLER Molecules Diffuse FASTER than LARGE Molecules.
MOST Short Distance Transport of Materials into and out of cells occurs by Diffusion.
Increase in PRESSURE will accelerate particle motion therefore diffusion.
- CONCENTRATION
- TEMPERATURE
- SIZE
- PRESSURE
- and the TYPE OF THE MOLECULES THAT IS DIFFUSING.
The more CONCENTRATED the substances, the more rapidly diffusion occurs because there are more collisions between the particles of a substances.
Molecules Diffuse FASTER at HIGHER TEMPERATURES than at lower temperatures.
SMALLER Molecules Diffuse FASTER than LARGE Molecules.
MOST Short Distance Transport of Materials into and out of cells occurs by Diffusion.
Increase in PRESSURE will accelerate particle motion therefore diffusion.
Concentration Gradient
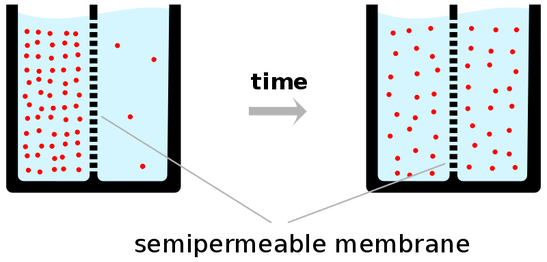
This difference in the Concentration of Molecules across a space is called a CONCENTRATION GRADIENT.
Diffusion will Eventually cause the Concentration of molecules to be the SAME throughout the space the molecules occupy.
When the Concentration of the molecules of a Substance is the Same Throughout a Space (no concentration gradient exists), a State of EQUILIBRIUM EXISTS
- DIFFUSION ALWAYS OCCURS DOWN A CONCENTRATION GRADIENT
- FROM THE AREA OF GREATER or HIGHER CONCENTRATION TO THE AREA OF LESS OR LOWER CONCENTRATION.
Diffusion will Eventually cause the Concentration of molecules to be the SAME throughout the space the molecules occupy.
When the Concentration of the molecules of a Substance is the Same Throughout a Space (no concentration gradient exists), a State of EQUILIBRIUM EXISTS
