Goal 2 - Organic Compounds
What is a Compound?
Compound is composed of two or more elements chemically combined.
Examples: Carbohydrates
Lipids
Proteins
Nucleic Acids
Carbon is the molecule of life…. CARBON = LIFE
TYPES OF ORGANIC COMPOUNDS
CARBOHYDRATES
1. Starch = branched glucose polymer
- fatty acid
Types of Lipids:
1. saturated – each carbon chain is bonded to other carbons by SINGLE bonds. (page 160)
2. unsaturated- each carbon chain is bonded to other carbons by a SINGLE DOUBLE bond (page 160)
3. polyunsaturated – each carbon in the chain is bonded to other carbons by MORE THAN ONE DOUBLE bond. (page 160)
Function of Lipids:
PROTEINS
Function of Proteins
NUCLEIC ACIDS
Types of Nucleic Acids
Lipids
Starch – Complex Carbohydrate (polysaccharide)
1. Iodine – Turns blue-black if starch is present.
Glucose – Simple Carbohydrate (monosaccharide)
1. Benedicts – Turns red-orange when heated if glucose is present.
Protein
1. Biurets – Turns lavender of a protein is present.
Compound is composed of two or more elements chemically combined.
Examples: Carbohydrates
Lipids
Proteins
Nucleic Acids
Carbon is the molecule of life…. CARBON = LIFE
TYPES OF ORGANIC COMPOUNDS
CARBOHYDRATES
- Provide energy
- Bimolecule composed of C (carbon), H (hydrogen) and O (oxygen)
- The ratio of C:H:O is 1:2:1
- Simplest type of carbohydrate is a “mono”saccharide – one sugar chain
- glucose C6H12O6
- fructrose
- “Di”saccharide – two sugar chains
- disaccharide = monosaccharide + monosaccharide
- sucrose = glucose + fructose
- “Poly”saccharide – many sugar chains
- polysaccharide = monosaccharide + monosaccharide + monosaccharide
1. Starch = branched glucose polymer
- Storage form of sugar in plants
- Storage form of glucose in animal’s liver
- Provides structure and support to plants (makes them stand upright)
- Bimolecule made of carbon (C), hydrogen (H), and Oxygen (O).
- Includes fats, oils, waxes and steroids.
- Insoluble in water “won’t mix or dissolve”
- No charge – “non”-polar – not attracted to water
- Composed of three fatty acids linked to a molecule of glycerol
- Lipid = Glycerol - fatty acids
- fatty acid
Types of Lipids:
1. saturated – each carbon chain is bonded to other carbons by SINGLE bonds. (page 160)
2. unsaturated- each carbon chain is bonded to other carbons by a SINGLE DOUBLE bond (page 160)
3. polyunsaturated – each carbon in the chain is bonded to other carbons by MORE THAN ONE DOUBLE bond. (page 160)
Function of Lipids:
- energy storage
- insulation
- protective coatings
- plasma membranes
PROTEINS
- Essential for Life
- Provide structure for tissues and organs
- Carry out metabolism
- Large, complex, polymers
- Composed of carbon (C), hydrogen (H), Oxygen (O), and Nitrogen (N)
- Building blocks are amino acids
- Amino acid + amino acid + amino acid = Protein
- Amino acids are bonded together (+) with Peptide bonds.
Function of Proteins
- Contracting muscle tissues
- Transport oxygen in the bloodstream (hemoglobin)
- Provide immunity
- Regulate other proteins
- Carry out chemical reactions à specialized protein called enzymes
NUCLEIC ACIDS
- Complex
- Stores all information in the form of codes
- Polymer made of nucleotides
- Nucleotide – nitrogen base (N) + simple sugar (CHO) + phosphate group (P)
Types of Nucleic Acids
- DNA – deoxyribosenucleic acid
- Copy organisms genetic code
- Has the instruction to form organism’s enzymes and structural proteins.
- Genetic code that determines how an organism will look and act.
- Passed on from one generation to the next.
- RNA – ribonucleic acid
- Forms a copy of DNA for use in making proteins.
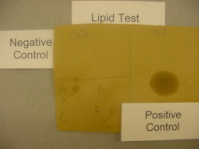
Lipids
- Brown Paper – Does it leave a greasy spot on brown paper? If so then, lipids are present.
- Alcohol solubility – Does it mix with alcohol? If so then, lipids are present.
Starch – Complex Carbohydrate (polysaccharide)
1. Iodine – Turns blue-black if starch is present.
Glucose – Simple Carbohydrate (monosaccharide)
1. Benedicts – Turns red-orange when heated if glucose is present.
Protein
1. Biurets – Turns lavender of a protein is present.